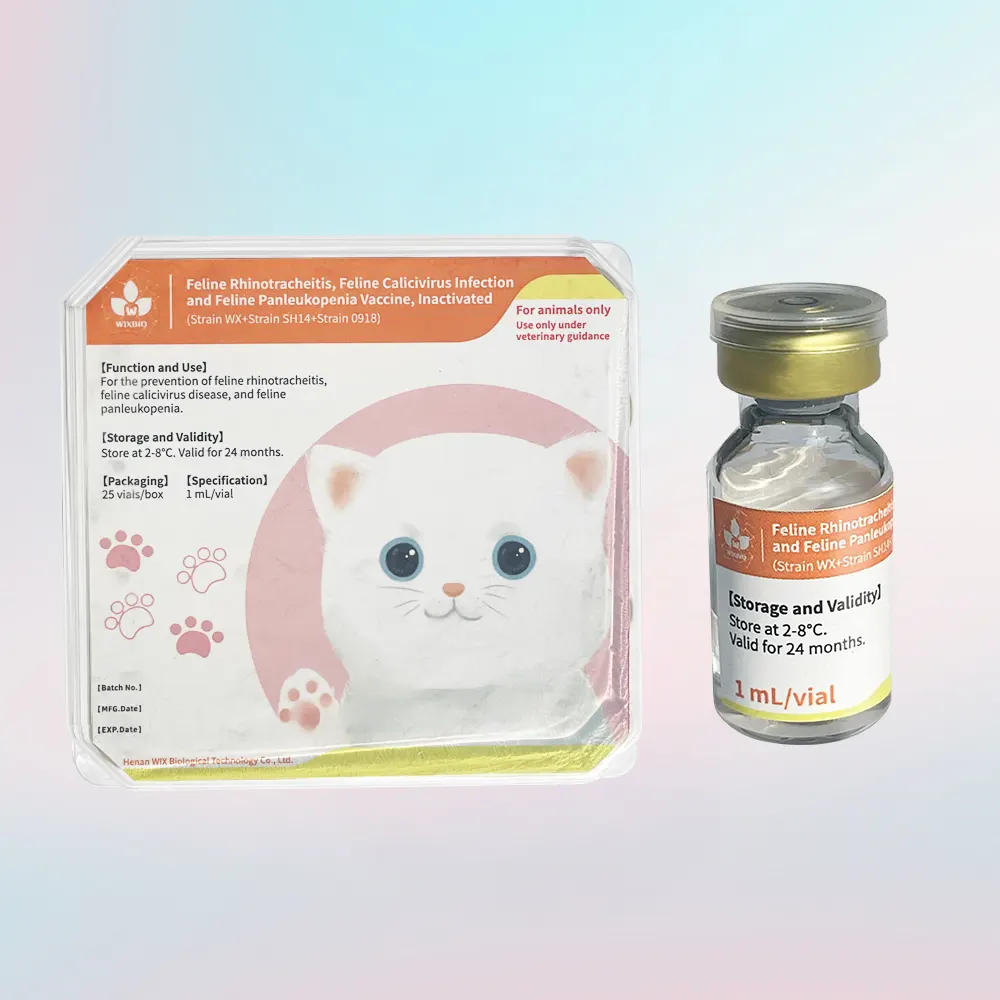
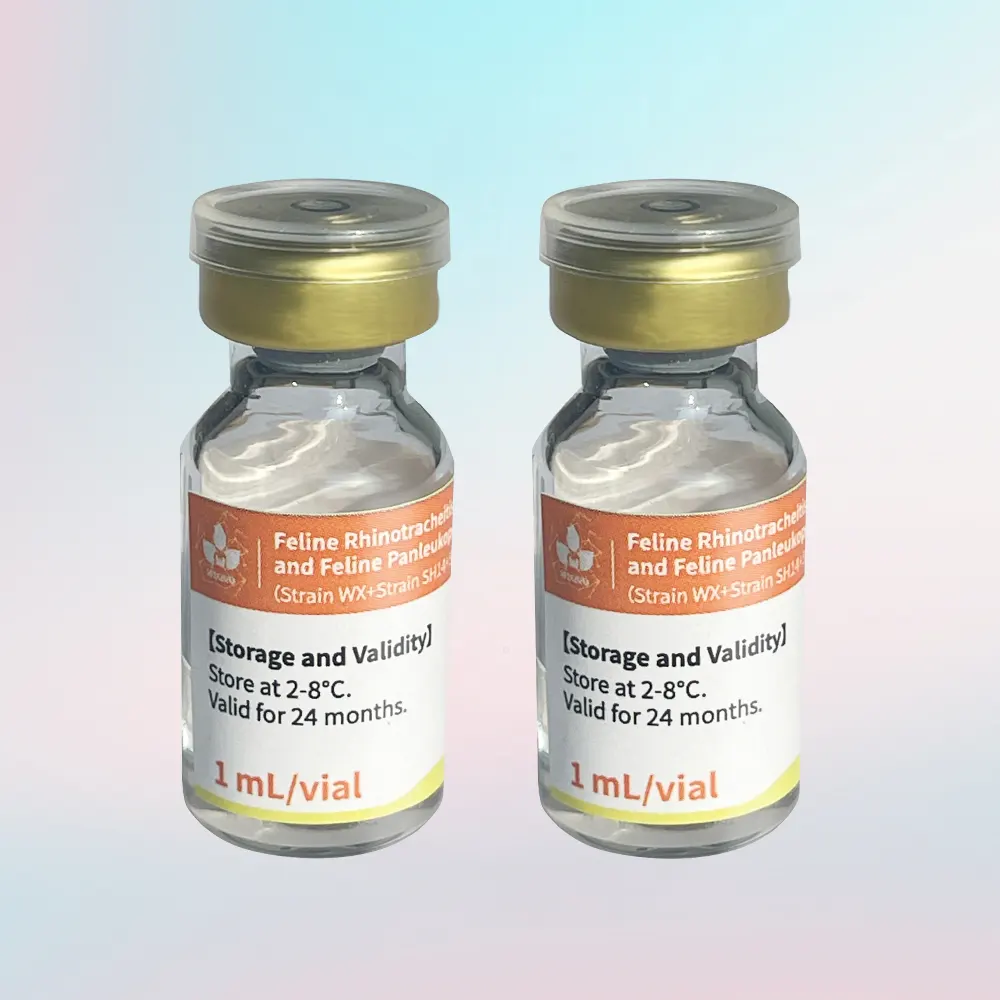
Feline Triple Vaccine Description
This inactivated combination vaccine is formulated to protect cats against three major infectious diseases: feline herpesvirus type 1 (FHV-1), feline calicivirus (FCV), and feline parvovirus (FPV).
Feline upper respiratory infections caused by FHV-1 and FCV are highly contagious and commonly seen in shelters, catteries, and multi-cat environments, leading to significant morbidity. Meanwhile, feline panleukopenia is a highly fatal disease, especially in kittens, characterized by severe leukopenia and gastrointestinal damage.
This vaccine contains well-selected strains (WX, SH14, and 0918) with high antigen content, ensuring strong immunogenicity while maintaining excellent safety due to its inactivated formulation.
Following the complete primary vaccination course, protective immunity develops within 10 days after the second dose, providing 12 months of effective protection. It is ideal for routine immunization programs in veterinary clinics, pet hospitals, and large-scale breeding operations.
Immunization Program
- Primary Vaccination (Kittens ≥ 8 weeks old):
- First dose: 1.0 mL
- Second dose: 1.0 mL after 21–28 days
- Booster Vaccination:
- Annual revaccination (1.0 mL)
Dosage & Administration
- Route: Subcutaneous injection
- Dosage: 1.0 mL per cat
Antigen Composition
Each 1.0 mL dose contains:
- Feline herpesvirus type 1 (FHV-1, strain WX): ≥ 107.0 TCID50 (before inactivation)
- Feline calicivirus (FCV, strain SH14): ≥ 108.0 TCID50
- Feline parvovirus (FPV, strain 0918): ≥ 104.0 TCID50
Safety Profile
- Generally no visible adverse reactions observed
- Rare allergic reactions may occur due to individual sensitivity
- In case of hypersensitivity:
- Immediate administration of epinephrine is recommended
- Supportive therapy as needed
Packaging & Specification
- Specification: 1 mL/vial
- Packaging: 25 vials/box
Storage & Shelf Life
- Storage: 2–8°C, protected from light
- Do NOT freeze
- Shelf Life: 24 months
Precautions
- Use only in healthy cats ≥ 8 weeks old
- Bring vaccine to room temperature and shake well before use
- Use immediately after opening
- Maintain aseptic injection procedures
- Dispose of used materials safely