Infectious bursal disease (IBD) in chickens, caused by infectious bursal disease virus (IBDV), is an acute, contact-transmissible disease primarily affecting 3- to 6-week-old chicks, leading to severe immunosuppression. In recent years, novel variant IBDV strains (nVarIBDV) have become widespread in China, with traditional vaccines showing limited efficacy. Based on 2025 surveillance data (from Qingdao Yibang Assurance Center and other institutions), WIXBIO addresses the “variable” (variant strain prevalence) and “constant” (environmental resistance, early infection risks) characteristics of IBDV, presenting comprehensive vaccine immunization and field control strategies, recommending the WIXBIO Trivalent IBD Vaccine to establish an effective control system.

1. IBDV Characteristics Analysis
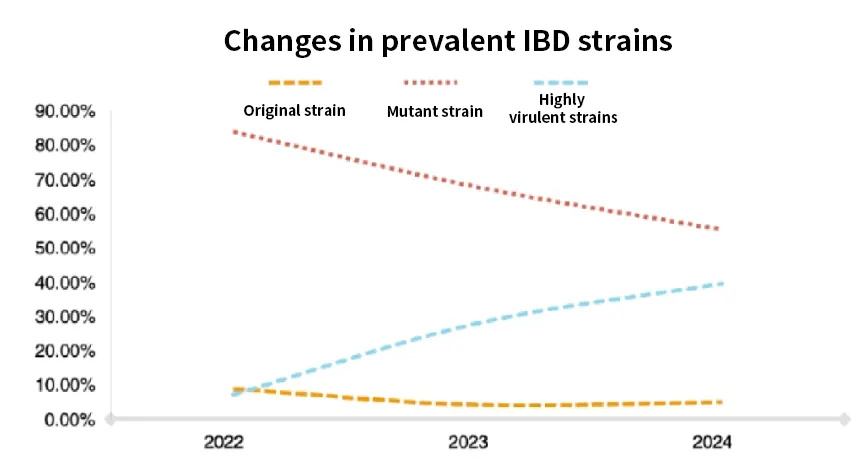
1.1 Variable: Viral Evolution Trends
- Evolutionary History: Evolved from classic virulent strains and very virulent IBDV (vvIBDV) to the currently dominant novel variant strains (nVarIBDV, A2dB1 genotype) and a minority of classic strains.
- 2025 Prevalence: Increased IBDV detection rates in white-feather broilers, young breeder chickens, and layer flocks, reported across 15 provinces (Northeast, East, and South China), with disease onset expanding to 15-35 days of age.
- Clinical Changes: Variant strains exhibit milder symptoms (primarily bursal swelling or atrophy) but induce more severe immunosuppression, reducing the efficacy of H5/H7 avian influenza and Newcastle disease vaccines by 30-40%.
1.2 Constant: Environmental Resistance and Risks
- Resistance: Non-enveloped virus, highly resistant to pH 2, 50°C heat, and disinfectants (e.g., quaternary ammonium compounds), surviving in poultry houses for up to 4 months.
- Early Infection Risks: Earlier infections (especially <14 days) cause greater B lymphocyte loss in the bursa of Fabricius, leading to severe immunosuppression; infections within 2 weeks are often overlooked.
2. Core Control Strategies
Based on IBDV characteristics, effective control relies on three indispensable measures:
- High Maternal Antibody Levels: Block early infections within the first 15 days.
- Stringent Biosecurity: Reduce exposure to wild-type viruses.
- Targeted Vaccination: Protect flocks in mid-to-late stages, using the WIXBIO Trivalent IBD Vaccine.
3. Field Control Measures
3.1 Selection of Chicks with High Maternal Antibodies
- Source: Select chicks from breeder flocks immunized 2-3 times (at least twice with inactivated nVarIBDV vaccines, e.g., A2dB1 strain), providing high antibody levels via yolk.
- Efficacy: Maternal antibody titers ≥1:1000 protect chicks for 14-21 days, reducing early infection rates by 50%.
- Case Study: A layer farm using high maternal antibody chicks reduced IBDV infection rates from 20% to 5% within 15 days.

3.2 Consistent Biosecurity Measures
- Cleaning and Disinfection:
- After the batch, thoroughly remove feces and feed residues. Disinfect with a 1% peracetic acid or 5% potassium monopersulfate powder spray, combined with 3% formaldehyde fumigation (24 hours, 20 g/m³).
- Fallow Period: Maintain a 7- to 14-day fallow period between batches to eliminate residual viruses.
- Personnel and Equipment Control:
- Personnel must shower and change into sterilized clothing (autoclaved at 121°C for 15 minutes); equipment (e.g., duck nets) must be soaked in 0.5% glutaraldehyde for 30 minutes.
- Vehicles and luggage are disinfected with ultraviolet light at the entrance (≥30 minutes, intensity >70 μW/cm²).
- Environmental Optimization: Maintain poultry house ventilation (ammonia level <10 ppm) and humidity (50-70%) to reduce the survival of wild viruses.
- Efficacy: A broiler farm implementing 3-4 cycles of biosecurity measures reduced IBDV detection rates from 15% to 2%.

3.3 Targeted Use of WIXBIO Trivalent IBD Vaccine
- Product Features: The WIXBIO Trivalent IBD Vaccine targets classic strains, vvIBDV, and nVarIBDV (A2dB1 genotype), incorporating recombinant VP2 subunits for broad-spectrum protection.
- Immunization Schedule:
- 1-7 Days of Age: Administer WIXBIO Trivalent Inactivated Vaccine, 0.3-0.5 mL per bird, via intramuscular (chest) or subcutaneous (neck) injection, pre-warmed to 25-30°C (30 minutes in 40°C water bath), suitable for chicks with high maternal antibodies.
- Regions with Variable Maternal Antibodies: Booster with WIXBIO attenuated live vaccine (e.g., B87 strain) after 14 days, 0.1-0.2 mL per bird, to minimize maternal antibody interference.
- Mid-to-Late Stage Protection:
- Administer a booster of WIXBIO Trivalent Inactivated Vaccine at 21-28 days to enhance long-term immunity.
- Protection rates reach 85-90%; in 2025, East China pilot farms achieved an average antibody titer of 1:256.
- Case Study: A Henan farm using WIXBIO Trivalent Vaccine and 7-day fallow disinfection reduced IBDV infection rates from 10% to 1%.
4. Immunization Protocols
- Storage: Refrigerate at 2-8°C; do not freeze. Use cold-chain transport (4°C).
- Pre-Warming: Warm in a 40°C water bath for 30 minutes, shaking every 5 minutes to ensure a temperature of 25-30°C.
- Injection: Ensure accurate single-dose administration, with a missed dose rate <1% and leakage/bleeding rate <2%; re-administer if necessary.
- Record-Keeping: Complete the “Immunization Record Form”; collect 20-30 serum samples 21 days post-vaccination for ELISA testing (target titer ≥1:128).
5. Special Recommendations for Variant IBDV Strains
- Sample Monitoring: Per Guidelines for Poultry Disease Sample Collection and Submission, collect bursa of Fabricius (1-2 g/bird) within 3 days of disease onset for RT-PCR detection of VP2 HVR (222T, 318D mutations) to assess wild virus pressure.
- Vaccine Adjustment: If antibody titers are <1:64, prioritize IBD Vaccine, optimized using Animal Cell Suspension Culture Technology.
- Enhanced Biosecurity: Disinfect equipment with 0.5% glutaraldehyde before and after immunization to prevent transmission of variant strains.
6. Case Studies
- Success Case: A broiler farm that adopted WIXBIO Trivalent Vaccine, utilized high maternal antibody chicks, and implemented biosecurity measures reduced IBDV infection rates from 10% to 1%, resulting in a 90% reduction in economic losses.
- Failure Case: A farm neglecting biosecurity and equipment disinfection experienced variant IBDV spread, with mortality rising to 8%.
7. Conclusion and Outlook
Effective IBD control requires a three-pronged approach: high maternal antibodies, stringent biosecurity, and the WIXBIO Trivalent IBD Vaccine. 2025 surveillance indicates an increasing prevalence of nVarIBDV, with heightened risks of traditional vaccine failure. Farms should integrate molecular diagnostics (RT-PCR), standardized operating procedures (SOPs), and WIXBIO’s advanced production technology while strengthening biosecurity to minimize immunosuppression and economic losses.
References:
- Fan L, et al. Novel variant infectious bursal disease virus suppresses Newcastle disease vaccination. Veterinary Microbiology, 2020.
- OIE, Manual of Diagnostic Tests for Terrestrial Animals, 2023.
- WIXBIO, Trivalent IBD Vaccine Development Report, 2025.